Publication Information
ISSN: 2641-6859
Frequency: Continuous
Format: PDF and HTML
Versions: Online (Open Access)
Year first Published: 2018
Language: English
| Journal Menu |
| Editorial Board |
| Reviewer Board |
| Articles |
| Open Access |
| Special Issue Proposals |
| Guidelines for Authors |
| Guidelines for Editors |
| Guidelines for Reviewers |
| Membership |
| Fee and Guidelines |
 |
Sports Recovery & Performance – Hyperbaric Oxygen Therapy
Malcolm R Hooper*
Clinical Director, OXYMED, 643 Chapel Street South Yarra, Victoria, Australia
Received Date: June 26, 2020; Accepted Date: July 02, 2020; Published Date: July 10, 2020
*Corresponding author: Malcolm R Hooper, Clinical Director, OXYMED, 643 Chapel Street South Yarra, Victoria, Australia. Tel: +61400044 955; Email: info@oxymed.com.au
Citation: Hooper MH (2020) Sports Recovery & Performance – Hyperbaric oxygen Therapy. Adv Ortho and Sprts Med: AOASM-126.
Sports Recovery & Performance
Elite athletes typically retire as a result of unresolved injury. The physical and mental toll of competing at the high-end level of sports culminates with the never-ending list of chronic injuries. Instead of enjoying their success, many athletes are invariably confronted with managing potentially serious health related challenges off the field of glory.
End of a Long Career
Performance, recovery, performance, recovery an endless cycle. Many elite athletes are in a chronic ‘cytokine storm’ – a prolonged phase of burn-out. International athletes are confronted with a range of metabolic challenges due to constant flights at altitude (reduced pressure and oxygen), time differences, chronic hypoxia, fatigue, lactic acid overload and sheer exhaustion. As tissue oxygenation diminishes (hypoxia) the secondary inflammatory changes and range of injuries increases dramatically. Tissues chronically exposed to hypoxia over express pro-inflammatory cytokine expressions. Over-expression of inflammatory cytokine gene clusters spread, potentiating autoimmune disorders [1].
Case 1: Before & After
Case 2: Before & After


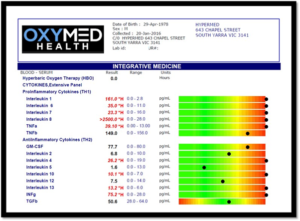
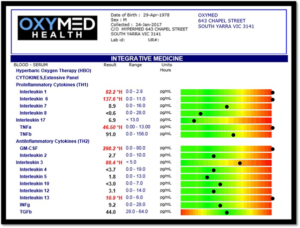
Cytokine Expressions
Cytokine are gene signal proteins and glycoproteins that ‘orchestrate’ proper immune responses including inflammation and anti-inflammatory pathways. Cytokines act as mediators and modulators and regulate immunological responses, hematopoietic development, and cell-to-cell communication as well as host responses to infectious agents and inflammatory stimuli. Cytokines are pleiotropic which refers to their ability to address multiple targets and physiological effects [1]. Cytokine production is tightly regulated, their homeostatic concentration in body fluids is low. However, if required, the concentration of cytokines can increase up to 1,000-fold [1]. In healthy individuals, cytokines are either not detectable or present at small concentrations in body fluid or tissues. Markers such as inflammatory cytokines are elevated after exercise in healthy individuals and return to baseline values within minutes to hours after exercise. Elevated concentrations of cytokines indicate activation of cytokine pathways associated with inflammation or disease progression. For this reason, cytokine measurements are both predictive and preventative to understand disease progression and monitor the effects of treatment.
Interleukin 1: (IL1) with IL6 and TNFα contribute to acute Cytokine Storm associated with COVID19 comorbidities [2]. IL1 is linked with systemic inflammation including ‘gut and brain connection’. Many athletes suffer with chronic irritable bowel syndrome (IBS). IL1 is linked in stroke with a reduction in the cerebral blood flow and increase in infarct volume [3]. Blockade of endothelin-1 receptors reversed this hypoperfusion, reduced tissue damage, and improved functional outcome. IL1 inhibits neurogenesis. IL1 is linked with synovial joint fluid degeneration and rheumatoid arthritis [4], IL1 is also linked in schizophrenia [5]. IL1 is linked in aggressive carcinoma progression including head and neck cancer, pancreatic cancer, thyroid cancer and bladder cancer [6].
Interleukin 6 (IL6): can increase up to a 1,000-fold during trauma and acute infection and is a major driver of COVID19 mortality [3]. IL6 is linked with chronic stress and may contribute to neurobehavioral complication [7]. IL6 is an important biomarker in monitoring inflammatory responses [8]. IL-6 is involved in the induction of acute phase responses and induction of fever [9]. IL6 is linked in chronic inflammatory arthritis and juvenile arthritis [10, 11]. Chronic elevation of serum IL-6 is associated with the progression of atherosclerosis in patients with vascular risk factors [11]. Elevated IL6 predicts cognitive decline and dementia [12]. IL6 is linked with chronic lumbar radicular pain. Persistent increase of IL1, IL6 and IL8 continues after disc herniation [9]. IL6 is linked with pathological pain associated with bone cancer, peripheral nerve injury, spinal cord injury, chemotherapy-induced peripheral neuropathy [8]. IL6 is a growth and survival factor in human glioblastoma cells and plays an important role in malignant progression.
Interleukin 8 (IL8): is linked with atherosclerosis and cerebrovascular disorders due to the micro-damage of the endothelial vascular wall [13]. IL8 is linked with activated microglia, as a primary source of neuroinflammation in concussion syndrome, head injury, alzheimers and other neurodegenerative diseases [14]. Hypoxia increased the release of IL8 which contributes to the aggressive spread of cancer cells [15].
Tumor Necrosis Factor Alpha: is a regulator of inflammatory responses. TNFα is linked with chronic pain syndromes, disc degeneration, failed surgery, arthritis, chronic tendinitis [16]. The hippocampus (role in learning and memory formation) is responsible for the growth and development of neurons (neurogenesis) after injury. TNFα is upregulated in the hippocampus and associated with neuropathic pain syndromes. TNFα suppresses brain-derived neurotrophic factor (BDNF) [17]. TNFα is linked with depression, neurodegeneration and maladaptive neuroplasticity.
S100B: is a marker of brain damage and BBB function. S100B is linked with depression disorders including dementia, alzheimers, stroke, traumatic brain injury, concussion, post traumatic stress disorders. S100B is associated with neural inflammation anywhere throughout the body [18, 19]. S100B is elevated in returning War Veterans suffering the effects of PTSD, TBI, and shock blast injuries. S100B elevation is associated with repeated hypobaric (long haul flights) hypoxic exposure resulting in blood-brain barrier dysfunction [20].
Hyperbaric Oxygen Therapy
HBOT is the application of two variables – increased pressure and increased oxygen. HBOT has profound effects on immune modulation in the presence of hypoxia. Mitochondrial respiration is the key to an athlete’s recovery and performance. HBOT studies demonstrate improvement in cerebral hypo perfusion, neuroinflammation, gastrointestinal inflammation, immune dysregulation, oxidative stress, mitochondrial dysfunction and neurotransmitter abnormalities [21]. HBOT is typically the focus of injury repair and part of an active rehabilitation program. All professional athletes and weekend warriors carry a growing injury list. Personalised protocols based on cytokine testing empower both predictive and preventative strategies. Off season strategies employing HBOT will best prepare an athlete and team. HBOT pre-conditioning off season, pre and post-game will diminish the window intersecting peak performance and the risk of injury and recurrence. HBOT increases the plasma saturation of oxygen. Normal plasma oxygen by volume is 3% (0.3 ml O2/100 ml blood), under HBOT conditions the O2 saturation increases 10-20 times that of breathing room air at normal atmospheric pressure [22]. HBOT increases the production and circulation of progenitor stem cells specific to the individual [23-25]. Oxygen saturated plasma under pressure ‘soaks the tissue’. HBOT saturation into the central nervous system promotes the brain, spinal cord and peripheral nerves to better regulate biochemical reactions [26]. HBOT promotes rapid recovery from soft tissue injuries. HBOT increased the number of proliferating and differentiating satellite cells, and the amount of regenerated muscle fibers [27]. HBOT inhibits chronic circulating infections in the blood plasma [28]. HBOT impacts underlying gut issues and sensitivities. All athletes suffer exposure to air borne pathogens, hypobaric hypoxic international flights and crowded environments [20]. HBOT has strong anti-inflammatory properties decreasing the pro-inflammatory cascade including IL1, IL6, IL7, IL8, TNFα, and S100B. HBOT has been shown to increase the counter-inflammatory IL4, IL10 and IL13 levels [22]. HBOT elevates IL10 and Brain Derived Neurotrophic Factor (BDNF) required for repetition skill neuroplasticity [18].
References
- Zhang JM, An J (2007) Cytokines, Inflammation and Pain. Int Anesthesiol Clin. Spring 45: 27–37.
- Jamilloux Y, Henry T, Belot A, Viel S, Fauter M, et al. (2020) Should We Stimulate or Suppress Immune Responses in COVID-19? Cytokine, Anti-Cytokine Interventions. J Rheumatol. 19:102567
- Lakhan SE, Kirchgessner A, Hofer M (2009) Inflammatory Mechanisms in Ischemic Stroke: Therapeutic Approaches. J Transl Med 17: 7-97.
- Nikfar S, Saiyarsarai P, Tigabu BM, Abdollahi M (2018) Efficacy and Safety of interleukin-1 Antagonists in Rheumatoid Arthritis. Rheumatol Int 38: 1363-1383.
- Shibuya M, Watanabe Y, Nunokawa A, Egawa J, Kaneko N, et al. (2014) Interleukin 1 Beta Gene and Risk of Schizophrenia: Updated Meta-Analysis. Hum Psychopharmacol 29: 31-37.
- Mantovani A, Barajon I, Garlanda C (2018) IL-1 and IL-1 Regulatory Pathways in Cancer Progression and Therapy. Immunol Rev 281: 57-61.
- Akioka S (2019) Interleukin-6 in Juvenile Idiopathic Arthritis. Mod Rheumatol 29: 275-286.
- Zhou YQ, Liu Z, Liu ZH, Chen SH, Li M (2016) Interleukin-6: An Emerging Regulator of Pathological Pain. J Neuroinflammation 13: 141.
- Moriya J (2019) Critical Roles of Inflammation in Atherosclerosis J Cardiol 73: 22-27.
- Niraula A, Witcher KG, Sheridan JF, Godbout JP (2019) Interleukin-6 Induced by Social Stress Promotes Anxiety. Biol Psychiatry 85: 679-689.
- Eltoft A, Arntzen KA, Wilsgaard T, Mathiesen EB, Johnsen SH, et al. (2018) Interleukin-6 Is an Independent Predictor of Progressive Atherosclerosis in the Carotid Artery. Atherosclerosis 271: 1-8.
- Bradburn S, Sarginson J, Murgatroyd CA (2018) Peripheral Interleukin-6 With Global Cognitive Decline in Non-demented Adults. Front Aging Neurosci 8: 9:438
- Apostolakis S, Vogiatzi K, Amanatidou V, Spandidos DA (2009) Interleukin 8 and Cardiovascular Disease. Cardiovasc Res 84: 353-360.
- McLarnon JG (2016) Chemokine Interleukin-8 (IL-8) in Alzheimer’s and Other Neurodegenerative Diseases. 6:273.
- Feldmeier J, Carl U, Carl H, Hartmann K, Sminia P (2003) yperbaric Oxygen does it promote growth or recurrence of malignancy (UHMS 2003) J Feldmeier 30: 1-18.
- Wang C, Yu X, Yan Y, Yang W, Zhagng H, et al. (2017) Tumor Necrosis Factor-α: A Key Contributor to Intervertebral Disc Degeneration. Acta Biochim Biophys 49:1-13.
- TNF-α Differentially Regulates Synaptic Plasticity in the Hippocampus and Spinal Cord by Microglia-Dependent Mechanisms After Peripheral Nerve Injury. J Neurosci 37: 871-881.
- Golden N, Mahadewa TGB, Aryanti C, Widyadharma IPE (2018) S100B Serum Level as a Mortality Predictor for Traumatic Brain Injury. Open Access Maced J Med Sci 6: 2239-2244.
- Arora P, Sagar R, Mehta M, Pallavi P, Sharma S (2019) Serum S100B Levels in Patients With Depression. Indian J Psychiatry 61: 70-76.
- Winter CD, Whyte J, Cardinal J, Kenny R, Ballard E (2016) Hypobaric Hypoxic Brain Injury of High Altitude: Plasma S100B Blood-Brain Barrier Dysfunction. Neurol Sci 37: 533-539.
- Paul Harch (2018) Oxygen and Pressure Epigenetics. Retrieved from: https://www.townsendletter.com/article/oxygen-and-pressure-epigenetics-understanding-hyperbaric-oxygen-therapy-after-355-years-as-the-oldest-gene-therapy-known-to-man/
- Golan H, Makogon B, Volkov O, Smolyakov Y, Hadanny A, et al. (2020) Imaging-based Predictors for Hyperbaric Oxygen Therapy Outcome in Post-Stroke Patients. Report 1. 136:109510.
- Increased circulating stem cells, cognitive performance in traumatic brain injury Subjects Following Hyperbaric Oxygen Therapy. Randomized Controlled Trial 44: 257-269.
- Thom SR, Bhopale VM, Velazquez OC, Goldstein LJ, Thom LH, et al. (2006) Stem cell mobilization by hyperbaric oxygen. Am J Physiol Heart Circ Physiol H1378-H1386.
- Tia PA, Chang CK, Niu KC, Lin MT, Chiu Wt, et al. (2010) Attenuating Experimental Spinal Cord Injury by Hyperbaric Oxygen: Stimulating Production of Vasculoendothelial and Glial Cell Line-Derived Neurotrophic Growth Factors and interleukin-10. J. Neurotrauma 27: 1121-1127.
- Jain KK (2004) Chapter 34 The role of HBO as ant in Rehabilitation and Sports Medicine: Textbook of Hyperbaric Medicine, 4th Edition, ed. Kewel K. Jain. Springer, Cham, Switzerland, 2004.
- Oyaizu T, Enomoto M, Yamamoto N, Tsuji K, Horie M, et al (2018) Hyperbaric Oxygen Reduces Inflammation, Oxygenates Injured Muscle, and Regenerates Skeletal Muscle via Macrophage and Satellite Cell Activation. Sci Rep 8:1288.
- Hooper MR (2018) Hyperbaric Medicine – The Life is in the Blood. Retrieved from: https://www.oxymed.com.au/books-by-malcolm-r-hooper