Publication Information
ISSN: 2641-7049
Frequency: Continuous
Format: PDF and HTML
Versions: Online (Open Access)
Year first Published: 2018
Language: English
| Journal Menu |
| Editorial Board |
| Reviewer Board |
| Articles |
| Open Access |
| Special Issue Proposals |
| Guidelines for Authors |
| Guidelines for Editors |
| Guidelines for Reviewers |
| Membership |
| Fee and Guidelines |
 |
NEUROPSYCHIATRIC DISORDERS and HEMOPYROLLACTAMURIA (HPU) – fact or fiction?
Erpenbach K1*, Erpenbach AS1, Kamsteeg2 J Seidl H3, Mayer W4, Baumeister-Jesch L5
1Institut für Leistungsoptimierung, Lange Heide 38, 50374 Erftstadt, Germany
2KEAC Parkstad, Snellius 1, 6422 RM Heerlen, Netherlands
3Immundiagnostik AG, Stubenwald-Alle 8a, 64625 Bensheim, Germany
4Lab4More GmbH, Augustenstraße 10, 80333 Munich, Germany
5HPU-Institut, In der Heidelslach 6, 69181 Leimen, Germany
Received Date: January 12, 2025; Accepted Date: February 12, 2025; Published Date: March 12, 2025;
*Corresponding author: Klaus Erpenbach, 1Institut für Leistungsoptimierung, Lange Heide 38, 50374 Erftstadt, Germany; Email: km.erpenbach@t-online.de
Abstract
Introduction: The term ‘Mauve factor’ (pyrroluria) dates back to 1958, when Dr Abram Hoffer defined this condition as a clinical disorder with elevated urine pyrrole levels: currently called hydroxyhaempyrrolin-2-on (HPL). It has been suggested that the increased urinary pyrrole excretion leads to a deficiency of zinc and vitamin B6 which has been increasingly observed in patients with psychiatric disorders such as schizophrenia, anxiety and depression. Until now, the only recommended treatment for hemopyrroluria (HPU) has been zinc and B6 supplementation. The aim of this study is to clarify open questions: Do HPU patients have typical anamnestic criteria? Is there only a deficiency of zinc and vitamin B6 or are other micronutrient deficiencies detectable? Is a standardised micronutrient supplementation therapeutical-ly effective?
Methods: 96 patients with psychiatric disorders (ADHD, depression, anxiety) were included in the study and their medical history were evaluated using a prospective standardized questionnaire. Following written informed consent, HPL in the 24-hour urine and various micronutrients were determined in the blood of the study participants before and after 6 months of standardized treatment with Bionovelia B complex, Bionovelia Q10R and Bionovelia K2/D3. The standardized questionnaire was re-evaluated after 6 months of treatment and the changes quantified. Wilcoxon, Pearson chi-square and Whitney U tests were used for statistical analysis. p≤0.05 was assumed to be significant and p≤0.01 highly significant.
Results: Of the 96 patients aged 43.48 ± 17.58 years (66 women/30 men), 52 (54.2%) suffered from depression, 28 (29.2%) from an anxiety disorder and 16 (16.6%) from ADHD. Typical additional anamnestic symptoms were fatigue (patients with depression / anxiety disorder / ADHD = 96% / 100% / 84%), susceptibility to infections (81% / 93% / 81%), irritable bowel syndrome (60% / 57% / 75%), insomnia (44% / 43% / 50%), Hashimoto’s thyroiditis (25% / 32% / 25%) and a family history of neuropsychiatric disorders [number in the family: 1(1-4) / 2(1-4) / 1(1-3)]. A urinary HPL excretion of 0.90 ± 0.41 µmol/l was determined in all patients. HPL excretion was significantly higher in men (man vs woman (m:f): 1.01±0.42 vs 0.85±0.39 mol/l – p=0.047). In the analysis of micronutrients, the levels of all bioavailable B vitamins [B1=34.96±7.61µg/l, B2=85.13±23.86µg/l, B6=5.19± 3.92µg/l, B9=7.93±4.77ng/ml, B12=245.82±162.30ng/ml) and 25(OH)-vitamin-D3 (29.98±13.14ng/ml) were decreased. The values of all trace elements (zinc, calcium, magnesium, copper, manganese, iron) in the whole blood were within the normal range in all patients. Q10 levels before treatment were within international reference ranges but not within therapeutic ranges of 1500-2500 µg/l. In men, whole blood levels of copper (m:f = 12.69±1.77 vs. 14.64±3.3µmol/l – p=0.003), manganese (m:f = 9.34±2.52 vs. 11.50±3.32µg/l – p=0.017) and calcium (m:f = 1.25±0.11 vs. 1.31±0.15mmol/l – p=0.019) were significantly lower than in women, while women had significantly lower whole blood iron levels (m:f = 464.58±57.70 vs. 423.81±39.27mg/l – p=0.0001) and serum Q10 levels (m:f = 961.83±378.98 vs. 837.93±333.87µg/l – p=0.022). Total HPL excretion over 24 hours was significantly negatively correlated with bioavailable vitamin B9 (-0.219, p=0.033) and negatively with whole blood calcium (-0.187, p=0.076) but significantly positively correlated with whole blood zinc (+0.257, p=0.015) and whole blood iron (+0.236, p=0.024). The total score (TS) formed from the anamnestic criteria showed a significant inverse correlation with whole blood selenium (-0.238, p=0.022), with whole blood zinc (-0.216, p=0.042) and with bioavailable vitamin B1 (-0.246, p=0.016). The TS was significantly reduced by the standardized micronutrient therapy (TS before vs. after treatment = 8.38±1.54 vs. 0.98±0.91 – p=0.0001). 22 of the 96 (23%) patients were receiving psychotropic drug therapy at the start of the study, which could be stopped during the 6-month micronutrient treatment. The 6-month micronutrient treatment led to a significant increase in all bioavailable B vitamins, vitamin D3, Q10, selenium, calcium and iron in whole blood.
Conclusion: Patients with neuropsychiatric disorders and increased HPL excretion show further typical symptoms that can be categorized into main criteria (fatigue, susceptibility to infections, neuropsychiatric disorders), secondary criteria (irritable bowel syndrome, insomnia, Hashimoto’s thyroiditis) and family criteria (number of first-degree family members affected by neuropsychiatric disorders). The resulting total score is very suitable for assessing curative micronutrient treatment. Contrary to the typical micronutrient deficiencies (zinc, magnesium, vitamin B6) in HPU patients described in the previous literature, 25(OH)-vitamin D3, all bioavailable B vitamins, whole blood selenium, whole blood calcium and whole blood iron are deficient in this study. Standardized micronutrient treatment was able to compensate for these deficiencies within 6 months, wean patients off psychotropic drugs and significantly reduce their overall symptoms. Case-control studies have to confirm the specificity and sensitivity of HPL determination in urine and randomized placebo-controlled treatment studies have to confirm the effectiveness of standardized micronutrient treatment in HPU patients.
Introduction
Pyrroluria (also known as cryptopyrrole, pyrrole or Mauve factor disease) is a controversially discussed disorder and is defined by the detection of elevated levels of pyrroles in the urine (1-4). Already in 1961, Irvine (1) was able to show that in patients with psychiatric disorders (depression, schizophrenia), substances are excreted in the urine that react with the Ehrlich reagent blue-purple and thus became known as ‘mauve factor’ (mauve = mauve-colored, light purple). The chromatographic properties of the ‘mauve factor’ corresponded to those of cryptopyrrole (2,4-dimethyl-3-ethylpyrrole) (3). It was assumed that the pyrrole possibly originated from a disturbed build-up or breakdown of hem and was an indicator of substances causing the psychiatric symptoms in the affected patients. Irvine revised this in 1978 (5), stating that the substance was probably not kryptopyrrole, but 2-hydroxyhemopyrrolen-5-one (hemopyrrollactam = HPL). C.C. Pfeiffer, the most important representative of the pyrrole hypothesis, developed a biochemical model according to pyrrole forms bounding irreversibly with pyridoxal-5-phosphate (active vitamin B6) and zinc (6). According to Pfeiffer et al (6), the increased excretion of pyrrole in the urine not only indicates a disorder of the hem metabolism but is also an indication of a loss of zinc and pyridoxal in affected patients, which causes the psychiatric symptoms. Active (bioavailable) vitamin B6 is a water-soluble vitamin and acts as an important coenzyme in various processes of iron metabolism, carbohydrate metabolism, amino acid metabolism, lipid metabolism and neurotransmitter synthesis (7). The HPL level increases under physical or psychosocial stress and is referred to as a ‘stress-induced factor’, which has already been confirmed with an experimental model (4,5). The neurotoxicity of HPL has also been repeatedly demonstrated in animal models (8). A theoretical mechanism of this neurotoxicity could be related to structural hem depression, but also to a similarity to pyroglutamate and kainic acid as well as their influence on neurotransmitter metabolism and intracellular mineral metabolic processes (4). However, an increase in HPL is also associated with genetic dysregulations in hem metabolism, neurotransmitter synthesis and the phosphitylation of B vitamins or oxidative stress caused by intoxication from heavy metals, toxic chemicals or malposition in the axial skeletal system (9).
The aim of this study is to clarify open questions: Do HPL patients have typical anamnestic criteria? Is there only a deficiency of zinc and vitamin B6 or are other micronutrient deficiencies detectable? Is a standardized micronutrient supplementation therapeutically effective?
Methods
96 patients with psychiatric disorders [ADHD (N=16), depression (N=52), anxiety disorder (N=28)] were included in the study and their medical history were evaluated using a new prospective standardized questionnaire (appendix). Symptoms with a frequency of > 80% were defined as main criteria and scored with 2 points each. Symptoms with a frequency < 80% were defined as secondary criteria and scored with one point each. Family criteria resulted from the declaration of first-degree family members with neuropsychiatric disorders and were scored with one point per declaration. The total score was determined by adding the main, secondary and family criteria. Following written informed consent, HPL in the 24-hour urine and various micronutrients were determined in the blood of the study participants before and after 6 months of standardized treatment with Bionovelia B complex, Bionovelia Q10R and Bionovelia K2/D3. The standardized questionnaire was re-evaluated after 6 months of treatment and the changes were quantified.
Hemopyrrollactam complex (HPL in urine)
The urine was collected in a vitamin C-buffered container for 24 hours after a defined portion had been passed, then 40 ml was sent to the laboratory at KEAC Parkstad, Netherlands, where it was determined using a spectrophotometric method following a sandwich immunoassay.
Whole blood trace elements selenium, zinc, copper, manganese, iron, calcium, magnesium (Na-heparin blood) vSe, vZn, vCu, vMn, vFe, vCa, vMg
Trace elements in whole blood were determined using induction-coupled mass spectrometry (ICP-MS) on a Perkin-Elmer platform (Nexion2000).
Coenzyme Q10 (serum)
Q10 in serum was determined using high-performance liquid chromatography (HPLC) with commercial kits (Chromsystems, Gräfelfing, Germany) according to the manufacturer’s instructions (Q10 – Order No. 68000).
Vitamin D 25-OH (serum)
The quantitative determination of total 25-OH vitamin D in serum was carried out according to the manufacturer’s instructions using a direct competitive chemiluminescence immunoassay (CLIA) on the Liasion-XL automated laboratory system from Diasorin, Dietzenbach, Germany (Order No. 310600).
Vitamin B1, B2, B6, B9, B12
The determination of vitamins B1, B2, B6, B9 (folic acid) and B12 was carried out using standardized microbiological test kits from the ID-Vit® product line (Immundiagnostik AG, Bensheim, Germany) in accordance with the manufacturer’s instructions. Specific microorganisms are added to the patient sample in a vitamin-specific deficiency medium. Quantification is carried out after incubation using a standard series.
B1 (EDTA whole blood): The whole blood was pretreated, diluted and added to microtiter plates containing Lactobacillus fermentum. Incubation was carried out at 37°C for 48 h.
B2 (serum): Serum samples were pretreated and pipetted onto microtiter plates containing Lactobacillus rhamnosus. Incubation was carried out at 37°C for 72 h.
B6 (serum): Serum was pretreated enzymatically and added to microtiter plates containing Saccharomyces cerevisiae. Growth was measured after 48 h incubation at 30°C.
B9 (folic acid, serum): Serum was pretreated and pipetted onto microtiter plates containing Lactobacillus rhamnosus. After 46-50 h incubation at 37°C the turbidity was determined.
B12 (serum): Serum was pretreated and added to microtiter plates containing Lactobacillus delbrueckii subsp. lactis. Incubation was carried out at 37°C for 46-50 h.
The growth of the microorganisms was quantified via turbidity at 620 nm. The method allows the determination of bioavailable vitamins.
All patients were treated with the standardized products of Fagus Pharma, Germany: BIONOVELIA B-complex capsules (1-0-1), BIONOVELIA Q10-R liquid (1 tablespoon in the morning), BIONOVELIA K2-200 µg/D3-10000 IE capsules (1-0-0) to be taken daily.
Statistical analyses were performed using the IBM Statistical Package for the Social Sciences (SPSS) 24 (IBM Corporation, Armonk, NY, USA) and Microsoft Excel 2016 (Microsoft Corporation, Redmond, WN, USA). The quantitative parameters were described by mean, median and minimum-maximum values. Pairwise comparisons were performed using the non-parametric Wilcoxon- and Mann-Whitney U-tests. The Pearson chi-square test was used to determine relationships between categorical variables. In all statistical tests, results were interpreted as statistically significant if p was less than 0.05, with p ≤ 0.01 considered highly significant.
Results
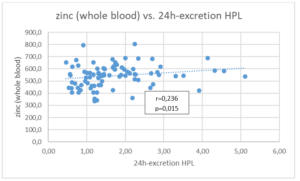
Of the 96 patients aged 43.48 ± 17.58 years (66 women/30 men), 52 (54.2%) suffered from depression, 28 (29.2%) from an anxiety disorder and 16 (16.6%) from ADHD. Typical additional anamnestic symptoms (Tab.1 ) were fatigue (patients with depression / anxiety disorder / ADHD = 96% / 100% / 84%), susceptibility to infections (81% / 93% / 81%), irritable bowel syndrome (60% / 57% / 75%), insomnia (44% / 43% / 50%), Hashimoto’s thyroiditis (25% / 32% / 25%) and a family history of neuropsychiatric disorders [number in the family: 1(1-4) / 2(1-4) / 1(1-3)]. The total score before treatment was 8.38±1.54 points and consisted of major criteria (5.52±0.86 points), minor criteria (1.34±0.88 points) and family criteria (1.55±0.78 points). There was no difference in the scores between women and men (Tab 1). A urinary HPL excretion of 0.90±0.41 µmol/l or 1.68±0.94 µmol/24h was determined in all patients. The HPL complex as well as the HPL excretion over 24h was significantly higher in men (man vs woman: – HPL complex: 1.01±0.42 vs 0.85±0.39 µmol/l – p=0.047; HPL 24h excretion: 1.97±1.04 vs 1.54±0.86 µmol/24h – p=0.029). In the analysis of micronutrients before treatment, the levels of all bioavailable B vitamins (B1=34.96±7.61µg/l, B2=85.13±23.86µg/l, B6=5.19±3.92 µg/l, B9=7.93± 4.77ng/ml, B12=245.82±162.30ng/ml) and 25(OH)-vitamin-D3 (29.98±13.14ng/ml) were reduced. Again, no difference was observed between women and men (Tab 2). The values of all trace elements (zinc, calcium, magnesium, copper, manganese, iron) in the whole blood and the Q10 levels before treatment were within the international reference ranges in all patients but Q10 not within the therapeutic range of 1500 – 2500 µg/l. In men, the whole blood values for copper (m:f = 12.69±1.77 vs. 14.64±3.3µmol/l – p=0.003), manganese (m:f = 9.34±2.52 vs. 11.50±3.32µg/l – p=0.017) and calcium (m:f = 1.25±0.11 vs. 1.31±0.15mmol/l – p=0.019) were significantly lower than in women, while women showed significantly lower iron levels in whole blood (m:f = 464.58±57.70 vs. 423.81±39.27mg/l – p=0.0001) and Q10 levels in serum (m:f = 961.83±378.98 vs. 837.93±333.87µg/l – p=0.022) (Tab.2 ). The total score (TS) formed from the anamnestic symptoms showed a significant inverse correlation with whole blood selenium (-0.238, p=0.022), with whole blood zinc (-0.216, p=0.042) and with bioavailable vitamin B1 (-0.246, p=0.016), but not with the HPL complex (0.017, p=0.870) or with total HPL excretion (0.017, p=0.870) or HPL total excretion over 24 hours (-0.059, p=0.565). However, the HPL complex correlates significantly with whole blood manganese (0.273, p=0.040) and highly significantly with total HPL excretion over 24 hours (0.632, p=0.0001) [Fig.1a-b]. Total HPL excretion over 24 hours correlated significantly negatively with bioavailable vitamin B9 (-0.219, p=0.033), indicated inversely with whole blood calcium (-0.187, p=0.076) and correlated significantly positively with whole blood zinc (+0.257, p=0.015) and whole blood iron (+0.236, p=0.024) [Fig.1c-e].
Table 1: Additive symptoms of the study participants related to the neuro-psychiatric diseases. N = number, ADHD = attention deficit hyperactivity disorder, VAS = visual analogue scale, IBS = irritative bowel syndrome
| ADHD N=16 | Depression N=52 | Anxiety N=28 | |
| MAIN CRITERIA (> 80% declarations) | |||
| fatigue (> VAS = 5) | 84% | 96% | 100% |
| susceptibility to infection (> twice per year) | 81% | 81% | 93% |
| SECONDARY CRITERIA (< 80% declarations) | |||
| IBS (all types with or without bloated belly) | 75% | 60% | 57% |
| insomnia (all types) | 50% | 44% | 43% |
| Thyreoiditis Hashimoto | 25% | 25% | 32% |
| FAMILY CRITERIA (number) | |||
| Relatives with known neuro-psychiatric disorders | 1 | 1 | 2 |
| (1-3) | (1-4) | (1-4) |
| Before treatment | After treatment | ||||||||
| all | female | male | p-value | all | female | male | p-value | p-value | |
| (N=96) | (N=66) | (N=30) | (m/f) | (N=96) | (N=66) | (N=30) | (m/f) | (before/after) | |
| Age | 43,48 ± 17,58 | 44,27 ± 16,80 | 41,4 ± 19,35 | 43,48 ± 17,58 | 44,27 ± 16,80 | 41,4 ± 19,35 | |||
| 0,365 | 0,365 | ||||||||
| (46,5: 7; 81) | (48: 11; 78) | (43,5: 7; 81) | (46,5: 7; 81) | (48: 11; 78 | (43,5: 7; 81) | ||||
| Total-Score | 8,38 ± 1,54 | 8,56 ± 1,34 | 7,97 ± 1,88 | 0,98 ± 0,91 | 0,95 ± 0,92 | 1,06 ± 0,91 | |||
| (≥5) | 0,151 | 0,529 | 0,0001 | ||||||
| (8: 5; 12) | (8: 6; 12) | (8: 1; 11) | (0,9: 0; 3,5) | (0,9: 0; 3,5) | (0,9: 0; 3,25) | ||||
| Main-Criteria | 5,52 ± 0,86 | 5,58 ± 0,82 | 5,4 ± 0,93 | 0,66 ± 0,78 | 0,61 ± 0,76 | 0,77 ± 0,83 | |||
| (≥4) | 0,352 | 0,317 | 0,0001 | ||||||
| (6: 4; 6) | (6: 4; 6) | (6: 4; 6) | (0,4: 0; 3) | (0,2: 0; 2,6) | (0,6: 0; 3) | ||||
| Secondary-Criteria | 1,34 ± 0,88 | 1,33 ± 0,92 | 1,37 ± 0,81 | 0,33 ± 0,49 | 0,35 ± 0,48 | 0,29 ± 0,47 | |||
| (≥1) | 0,769 | 0,758 | 0,0001 | ||||||
| (1: 0; 3) | (1: 0; 3) | (1: 0; 3) | (0; 0; 1,5) | (0: 0; 1,5) | (0: 0; 1,25) | ||||
| Family- | 1,55 ± 0,78 | 1,64 ± 0,85 | 1,37 ± 0,56 | no change | no change | no change | |||
| Criteria | 0,199 | ||||||||
| (≥1) | (1: 1; 4) | (1: 1; 4) | (1: 1; 3) | ||||||
| HPU-complex | 0,90 ± 0,41 | 0,85 ± 0,39 | 1,01 ± 0,42 | not determined | not determined | not determined | |||
| (standard < 0,8 µmol/l) | 0,047 | ||||||||
| (0,84: 0,21; 2,31) | (0,78: 0,21; 2,31) | (0,94: 0,42; 2,28) | |||||||
| volume 24h-urine | 1,93 ± 0,76 | 1,90 ± 0,78 | 1,97 ± ,72 | not determined | not determined | not determined | |||
| (1,1-2,1l) | 0,591 | ||||||||
| (1,88: 0,66; 4.70) | (1,88: 0,7; 4,70) | (1,85: 0,66; 3,05) | |||||||
| HPL- 24h-excretion | 1,68 ± 0,94 | 1,54 ± 0,86 | 1,97 ± 1,04 | not determined | not determined | not determined | |||
| (µmol/24h) | 0,029 | ||||||||
| (1,43: 0,46; 5,11) | (1,30: 0,54; 5,11) | (1,74: 0,46; 4,56) | |||||||
| zinc (VB) | 543,73 ± 91,65 | 536,56 ± 87,73 | 557,83 ± 98,91 | 552,68 ± 70,97 | 548,36 ± 67,47 | 561,14 ± 77,97 | |||
| [standard: 400 – 750 µg/dl] | 0,098 | 0,353 | 0,305 | ||||||
| (551: 334; 805) | (546: 334; 805) | (570: 340; 688) | (554: 339; 681) | (554: 339; 679) | (554,5: 356; 681) | ||||
| copper (VB) | 13,88 ± 2,95 | 14,64 ± 3,3 | 12,69 ± 1,77 | 13,49 ± 2,74 | 14,21 ± 3,43 | 12,73 ± 1,51 | |||
| [standard: 11 – 28 µmol/l] | 0,003 | 0,111 | 0,625 | ||||||
| (13,4: 10,3; 29,1) | (14,2: 10,3; 29,1) | (12: 10,5; 16,6) | (12,9: 10; 26,1) | (13,4: 10; 26,1) | (12,6: 10,4; 16) | ||||
| manganese (VB) | 10,67 ± 3,20 | 11,50 ± 3,32 | 9,34 ± 2,52 | 11,62 ± 3,37 | 12,44± 3,53 | 10,77 ± 3,05 | |||
| [standrad: 4,8 – 18 µg(l] | 0,017 | 0,127 | 0,102 | ||||||
| (10,3: 4,9; 17,1) | (11,7: 5,7; 17,1) | (9,5: 4,9; 14,4) | (11,7: 4,8; 19) | (12,4: 6,4; 19) | (10,70: 4,8; 17,3) | ||||
| Selen (VB) | 130,20 ± 21,52 | 130,42 ± 20,73 | 129,72 ± 23,44 | 150,18 ± 24,81 | 149,77 ± 24,96 | 151,06 ± 24,87 | |||
| [Norm: 121 -168 µg/l] | 0,837 | 0,783 | 0,0001 | ||||||
| (126: 84; 193) | (126,1: 84; 193) | (126: 93,6; 176) | (152,50: 104,5; 228) | (148,05: 105,4; 228) | (155,5: 104,5; 197) | ||||
| calcium (VB) | 1,29 ± 0,14 | 1,31 ± 0,15 | 1,25 ± 0,11 | 1,39 ± 0,13 | 1,42 ± 0,12 | 1,33 ± 0,12 | |||
| [standard: 1,14 – 1,68 mmol/l] | 0,019 | 0,003 | 0,0001 | ||||||
| (1,27: 0,9; 1,7) | (1,31: 0,9; 1,7) | (1,23: 1,08; 1,51) | (1,39: 1,1; 1,7) | (1,4: 1,2; 1,7) | (1,33: 1,1; 1,56) | ||||
| magnesium (VB) | 1,38 ± 0,12 | 1,37 ± 0,12 | 1,39 ± 0,12 | 1,39 ± 0,11 | 1,38 ± 0,11 | 1,42 ± 0,09 | |||
| [standard: 1,29 – 1,69 mmol/l] | 0,231 | 0,168 | 0,163 | ||||||
| (1,35: 1,19; 1,68) | (1,34: 1,19; 1,68) | (1.37: 1,19; 1,64) | (1,38: 1,1; 1,6) | (1,37: 1,1; 1,6) | (1,39: 1,24; 1,59) | ||||
| ferrum (VB) | 437,25 ± 49,73 | 423,81 ± 39,27 | 464,58 ± 57,70 | 455,51 ± 35,65 | 442,62 ± 26,06 | 482,18 ± 38,35 | |||
| [standard: 420 -460 mg/l] | 0,0001 | 0,0001 | 0,001 | ||||||
| (436: 326; 554) | (421: 326; 501) | (474,5: 344; 554) | (451: 366; 576) | (443: 366; 498) | (467: 429,5; 576) | ||||
| thiamine [B1] (bioavailable) | 34,96 ± 7,61 | 34,63 ± 7,8 | 35,69 ± 7,24 | 60,31 ± 20,91 | 58,63 ± 21,46 | 64,02 ± 19,49 | |||
| [standard > 40 µg/l] | 0,237 | 0,154 | 0,0001 | ||||||
| (32: 23; 61) | (31,85: 23; 61) | (34: 23; 56.5) | (54,6: 27; 156,2) | (53,75: 27; 156,2) | (57,35: 42,1; 108,4) | ||||
| riboflavin [B2] | 85,13 ± 23,86 | 82,55 ± 19,82 | 90,83 ± 30,60 | 131,43 ± 62,73 | 128,75 ± 62,52 | 137,33 ± 63,85 | |||
| (bioavailable) | 0,302 | 0,583 | 0,0001 | ||||||
| [Norm > 90 µg/l] | (81: 49; 213) | (81: 49; 180) | (81,7: 55,4; 213) | (110,55: 50; 442) | (110,15: 50; 442) | (113,7: 73,9; 307) | |||
| Pyridoxal [B6] | 5,19 ± 3,92 | 5,01 ± 4,09 | 5,57 ± 3,56 | 46,11 ± 27,98 | 46,97 ± 30,58 | 44,21 ± 21,51 | |||
| (bioverfügbar) | 0,416 | 0,994 | 0,0001 | ||||||
| [standard > 10 µg/l] | (3,6: 2; 30) | (3,6: 2; 30) | (3,6: 3; 15,5) | (35,05: 3,6; 131,6) | (35,05: 3,6; 131,6) | (31,9: 18,9; 86,5) | |||
| folic acid [B9] | 7,93 ± 4,77 | 8,12 ± 4,36 | 7,52 ± 5,60 | 18,71 ± 12,57 | 20,34 ± 14,52 | 15,11 ± 5,04 | |||
| (bioavailable) | 0,128 | 0,219 | 0,0001 | ||||||
| [standard > 9 ng/ml] | (6,8: 2,1; 31,6) | (7,2: 2,1; 28,2) | (5,4: 2,5; 31,6) | (15,9: 4,8; 93,1) | 16,05: 5; 93,1) | (15,15: 4,8; 23) | |||
| cobalamin [B12] | 245, 82 ± 162,30 | 243,51 ± 164,64 | 250,82 ± 159,75 | 457,98 ± 347,66 | 425,06 ± 287,91 | 530; 39 ± 449,72 | |||
| (bioavailable) | 0,776 | 0,246 | 0,0001 | ||||||
| [standard > 400 ng/ml] | (219: 24,5; 599) | (189: 24,5; 599) | (253,15: 59,8; 578) | (526,5: 87,3; 2368) | (509,6: 87,3; 1030) | (571,3: 119; 2368) | |||
| VitaminD3 | 29,98 ± 13,14 | 30,48 ± 11,36 | 28,87 ± 16,52 | 93,65 ± 43,21 | 99,39 ± 43,05 | 81,19 ± 41,57 | |||
| (standard: 30 – 80ng/ml] | 0,151 | 0,024 | 0,0001 | ||||||
| (27,5: 7,3; 90,7) | (27,8: 7,3; 71,5) | (24,4: 10,9; 90,7) | (78,8: 26,3; 230) | (89,3: 35,8; 230) | (63,9: 26,3; 195) | ||||
| Qu10 | 837,93 ± 333,87 | 780,74 ± 296,86 | 961,83 ± 378,98 | 2191,12 ± 1188,91 | 2251,76 ± 1240,85 | 2057.70 ± 1073,60 | |||
| [standard: 750 – 1200 µg/l] | 0,022 | 0,690 | 0,0001 | ||||||
| (790: 284; 1918) | (701: 284; 1538) | (942,5: 444; 1918) | (1855: 629; 5710) | (1937,5: 629; 5710) | (1843,5: 740; 5276) | ||||
Table 2: Demographic characteristics (participants). N = number, (median: minimum; maximum). M = Male / W = Female – HPL = Hemopyrrolactam, VB = Whole blood
Figure 1a: Correlation between whole blood manganese (µg/l) and the HPL complex (in µmol/l), l = liter.
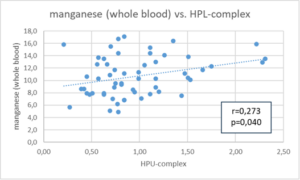
Figure 1b: Correlation between the total excretion of hemopyrrolactam (HPL in µmol/24h) and the HPL complex (in µmol/l), h = hours, l = liter.
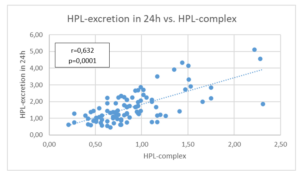
Figure 1c: Correlation between bioavailable vitamin B9 (folic acid in ng/ml) and the total excretion of hemopyrrollactam (HPL in µmol/24h), h = hours.
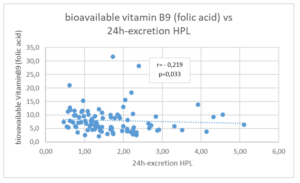
Figure 1d: Trend between whole blood calcium (mmol/l) and the total excretion of hemopyrrollactam (HPL in µmol/24h), h = hours, l = liter.
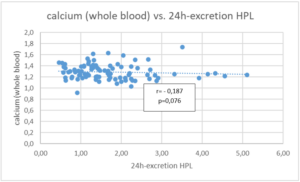
Figure 1e: Correlation between whole blood zinc (µg/ml) and the total excretion of hemopyrrollactam (HPL in µmol/24h), h = hours.
TS was significantly reduced by the standardized micronutrient treatment for 6 months (TS before vs. after therapy = 8.38±1.54 vs. 0.98±0.91 – p=0.0001). 22 of the 96 (23%) participants were receiving psychotropic drug therapy at the start of the study (mirtazapine N=12, opipramol N=7, escitalopram N=2, venlafaxine N=1), which could be stopped during the 6-month micronutrient treatment. The 6-month micronutrient treatment led to a highly significant increase in all bioavailable B vitamins (B1 before vs. after: 34.96±7.61 vs. 60.31±20.91 µg/l – p=0.0001, B2: 85.13±23.86 vs 131.43±62.73 µg/l – p=0.0001, B6: 5.19±3.92 vs 46.11±27.98 µg/l – p=0.0001, B9: 7.93±4.77 vs 18.71±12.57 ng/ml – p=0.0001, B12: 245.82±162.30 vs 457.98±347.66 ng/ml -p=0.0001), of vitamin D3 (29.98±13.14 vs 93.65±43.21 ng/ml – p=0.0001), of Q10 (837.93±333.87 vs 2191.12±1188.91 µg/l – p=0.0001), of selenium (130.20±20 vs 150.18±24.81 µg/l – p=0.0001), calcium (1.29±0.14 vs 1.39±0.13 mmol/l – p=0.0001) and iron (437.25±49.73 vs 455.51±35.65 mg/l – p=0.001) in whole blood. Copper, manganese and zinc in whole blood remained unchanged under micronutrient treatment (Tab 2). No side effects were reported.
Discussion
There are no international studies on the evaluation of further symptoms in patients with neuropsychiatric diseases and increased HPL excretion in urine. Chronic susceptibility to infections, especially against viruses (mainly against herpes viruses) and progressive fatigue were the most frequently mentioned additive symptoms in our study, regardless of the neuro-psychiatric diagnosis; irritable bowel symptoms, sleep disorders and symptoms of thyroid dysfunction were further symptoms. What is striking – but also emphasizes the genetic origin of HPU – is the cross-generational appearance of first-degree family members with neuropsychiatric disorders (Tab.1). These symptoms can be used to create a catalogue of criteria which at first – if fulfilled – has led to the determination of HPL excretion over 24 hours and an overall score can be calculated in a reproducible manner, making it possible to compare the results in future studies (Fig. 2).
Figure 2: HPU-Criteria

However, given the long history of identifying elevated HPL and neuropsychiatric disorders, the reproducibility of HPL determination remains controversial (1-6). Also, the biochemical origin of HPL and the mechanism for increased HPL excretion is unknown. According to the review of Warren et al. in 2021, numerous studies have attempted to show a relationship between elevated pyrrole levels and the diagnosis of neuropsychiatric disorders, but with limited impact (10). Unfortunately, most of these studies did not use statistical analyses and/or control group data or had limited sample sizes, which limited the validity of the data (6,11-19). Only the Fryar-Williams study showed a significant correlation between urinary pyrrole concentration and symptoms of schizoaffective psychosis or schizophrenia patients (20). In our study, an increased urinary HPL excretion of 0.90±0.41 µmol/l or 1.68±0.94 µmol/24h (according to the patented method of KEAC Parkstad) was determined in all patients with a neuro-psychiatric disorder (ADHD, depression, anxiety disorder). However, this method of determining HPL excretion (KEAC Parkstad) does not correlate with the total score (formed from all patient symptoms) and exceeded the international limit (15µg/l or 1µmol/l) in only 13.5% of cases. However, the concentration of the HPL complex correlates highly significantly with the total HPL excretion over 24 hours and with various micronutrients (folic acid, manganese, calcium, iron, zinc). Since Pfeiffer (6), one of the most important representatives of the pyrrole hypothesis, who developed the biochemical model according to pyrroles connecting irreversibly with pyridoxal-5-phosphate and zinc resulting in pyridoxal and zinc deficiency causes the neuro-psychiatric symptoms of the affected patients, no further studies have been done to reveal the involvement of additional micronutrients in neuropsychiatric disorders with increased HPL excretion. In our study, the concentrations of all bioavailable B vitamins (B1, B2, B6, B9, B12) and 25(OH)-vitamin D3 were lowered in the micronutrient analysis prior to treatment. However, the concentrations of all trace elements (zinc, calcium, magnesium, copper, manganese, iron) in the whole blood and the Q10 value were within the normal range in all patients before treatment. Mikirova (19) was also unable to detect any trace element deficiencies (copper, manganese, zinc, iron, selenium) in the whole blood of her patients with neuropsychiatric disorders (ADHD, schizophrenia, bipolar disorder, depression) and increased HPL excretions. Jackson et al (21) found a vitamin C deficiency and lowered zinc levels, but normal magnesium concentrations in whole blood in their patients with ADHD and increased HPL excretion.
The total score (TS) calculated from all symptoms of our study participants showed a significant inverse correlation with their selenium (-0.238, p=0.022) and zinc levels in whole blood (-0.216, p=0.042) as well as with their bioavailable vitamin B1 levels (-0.246, p=0.016). Total HPL excretion over 24 hours correlated significantly negatively with bioavailable vitamin B9 (-0.219, p=0.033), indicated an inverse correlation with calcium in whole blood (-0.187, p=0.076) and correlated significantly positively with zinc (+0.257, p=0.015) and iron concentrations in whole blood (+0.236, p=0.024). These results suggest that treatment with pyridoxal-5-phosphate, zinc and magnesium alone is not appropriate and explain the contradictory results in the treatment of patients with neuropsychiatric disease and increased HPL excretion (22). In our study, standardized micronutrient treatment significantly reduced TS (TS before vs. after therapy = 8.38±1.54 vs. 0.98±0.91 – p=0.0001). The patients who received psychotropic drug therapy before treatment were able to stop the psychotropic drugs during the 6-month micronutrient treatment. The standardized micronutrient treatment led to a highly significant increase in all bioavailable B vitamins, 25(OH)vitamin D3, Q10 and the trace elements selenium, calcium and iron in whole blood. Copper, manganese and zinc in whole blood remained unchanged under micronutrient therapy. There are no comparable studies available internationally. Taking these results into account, the effect of micronutrient treatment explains both: the genetic and acquired origin of HPU.
Voisey et al and Wockner et al have identified genetic markers (DTNBP1, DISC 1) associated with neuropsychiatric disorders and oxidative stress in several studies (23-26). It is therefore very likely that there are genetic and environmental factors that lead to redox dysregulation and manifest themselves in the form of elevated urinary pyrrole levels (9). Rambaud et al. confirmed that oxidative stress is not only associated with schizophrenia but also with other neuropsychiatric and metabolic disorders (27). Our own genetic studies in patients with neuropsychiatric disorders (ADHD, depression, anxiety disorder) and increased HPL excretion in urine show the following changes: in decreasing frequency COMT (N=4), MTHFR (N=3), TPH1 (N=2), MAOA (N=2), IL6 (N=1), eNOS (N=1) and thus also indicate the possibility of an inflammatory-oxidative process (own unpublished data). COMT (catecholamine O-methyl transferase), which is primarily responsible for the rapid degradation of dopamine and noradrenaline and requires various cofactors (B1, B2, B6, B9, B12, Q10), leads to panic disorders in the active form VAL158, particularly in women (28).
Genes of tryptophan hydroxylase (TPH), the limiting enzyme of serotonin synthesis, which catalyzes the hydroxylation of tryptophan to 5-hydroxytryptophan (5-HTP) and requires vitamin B9 (folic acid), magnesium and vitamin C as a cofactor, favor the development of depressive disorders in the case of genetic changes (29). Monoamine oxidase A (MAOA), the determining enzyme of serotonin metabolism, requires vitamin B2 as a cofactor and leads to aggressive behavior in the case of genetic changes (30). The interleukin-6 (IL6) and eNOS polymorphisms emphasize the component of oxidative stress as a trigger of neuropsychiatric disorders and require a high antioxidant capacity of selenium, zinc, vitamin C and Q10 as counterparts. Common to all genetic triggers is the dependence on all bioavailable B vitamins and trace elements. The generalized deficiency of all bioavailable B vitamins and Q10 in our study and the significant dependence on symptoms and HPL excretion over 24 hours as well as the elimination of symptoms by optimizing all micronutrients under treatment are consistent with both genetics and oxidative radical processes.
There are numerous open questions that should be clarified with further research: Firstly, the chemistry of pyrrole – does it need to be stabilized and frozen for transport and protected from light? Does this make chromatography mass spectrometry (LC/MS) the gold standard for detecting pyrrole in urine? Secondly, the origin of the molecule needs to be investigated and narrowed down. Case-control studies are suitable for this. Thirdly, the discrepancy between the serological B vitamin levels in serum (inactive form) and the bioavailable B vitamins (active form) as well as the trace elements in serum and whole blood also need to be clarified in case-control studies. Fourthly, are micronutrients increasingly bound and excreted via the HPL complex (a case-control study would also be suitable for this)? Fifthly, the clarification of genetics (neurotransmitters, oxidative stress, B vitamin metabolism) in the development of HPU. Sixth: can the application of symptom categories be used to distinguish between genetic and acquired origins and which triggers can be identified in acquired HPU? Seventh, double-blind, placebo-controlled studies are needed to prove the efficacy of micronutrient treatment in cases of neuro-psychiatric disorders.
As to the limitations of our study, it did not include healthy controls for comparison. In spite of the moderate number of cases, we decided not to execute P adjustment (e. g., Bonferroni, Bonferroni-Holm, or Hochberg procedures) for multiple testing. Thus, some of our findings might be due to chance. Eventually, the study was conducted in a mixed-cases cohort of 3 different neuro-psychiatric disorders, which may limit our conclusions.
In summary, there is clear and replicable clinical evidence that increased urinary HPL excretion is associated with neuropsychiatric distress. Without further research that validly clarifies all of the above questions, it is not possible to assess whether hemopyrroluria in patients with neuro-psychiatric disorders is a fact or fiction.
References
- Irvine D. Apparently non-indolic Ehrlich-positive substances related to mental illnesses. J Neuropsychiatry 1961:292–305
- Hoffer A. The discovery of kryptopyrrole and its importance in diagnosis of biochemical imbalances in schizophrenia and in criminal behavior. J Orthomol Med 1995;10:3–6
- Irvine D, Bayne W, Miyashita H. Identification of kryptopyrrole in human urine and its relation to psychosis. Nature 1969;224(5221):811–883
- McGinnis W, Audhya T, Walsh W, et al. Discerning the mauve factor, part 1. Altern Ther Health Med 2008;14:40–50
- Irvine D. Hydroxy-hemopyrrolenone, not kryptopyrrole, in the urine of schizophrenics and porphyrics. Clin Chem 1978;24:2069–2070
- Pfeiffer CC, Sohler A, Jennery C, et al. Treatment of pyroluric schizophrenica with large doses of pyridoxine and a dietary supplement of zinc. J Orthomol Psychiatr 1974;3:292–300
- Vrolijk MF, Opperhuizen A, Jansen EHJM, Hageman GJ, Bast A, Haenen GRMM. The vitamin B6 paradox: Supplementation with high concentrations of pyridoxine leads to decreased vitamin B6 function. Toxicology in Vitro 2017;44:206-212
- Cutler MG, Graham DJ, Moore MR. The mauve factor of porphyria, 3-ethyl-5-hydroxy-4,5-dimethyl-delta-3-pyrroline-2-one: Effects on behaviour of rats and mice. Pharmacol. Toxicol. 1990; 66(1): 66–68
- Lambert B, Semmler A, Beer C, Voisey J. Pyrroles as a Potential Biomarker for Oxidative Stress Disorders. Int J Mol Sci 2023;24:2712-2719
- Warren B, Sarris J, Mulder RT, Rucklidge JJ. Pyroluria: Fact or Fiction? J. Altern. Complement. Med. 2021, 27, 407–415
- Hoffer A. Malvaria, schizophrenia and the HOD test. Int. J. Neuropsychiatr. 1966, 2, 175–178
- Hoffer A, Mahon M. The presence of unidentified substances in the urine of psychiatric patients. J. Neuropsychiatr. 1961, 2, 331–362
- O’Reilly PO, Hughes G, Russell, RT, Ernest, M. The mauve factor: An evaluation. Dis. Nerv. Syst. 1965, 26, 562–568
- Sohler, A, Beck, R, Noval, JJ. Mauve factor re-identified as 2,4-dimethyl-3-ethylpyrrole and its sedative effect on the CNS. Nature 1970, 228, 1318–1320
- Walsh, WJ, Glab, LB, Haakenson, ML. Reduced violent behavior following biochemical therapy. Physiol. Behav. 2004, 82, 835–839
- Stuckey, R, Walsh, W, Lambert, B. The Effectiveness of Targeted Nutrient Therapy in Treatment of Mental Illness. J. Australas. Coll. Nutr. Environ. Med. 2010, 29, 3
- Cruz, R, Vogel, WH. Pyroluria: A poor marker in chronic schizophrenia. Am. J. Psychiatry 1978, 135, 1239–1240
- Gorchein, A. Urine concentration of 3-ethyl-5-hydroxy-4,5-dimethyl-delta 3-pyrrolin-2-one (‘mauve factor’) is not causally related to schizophrenia or to acute intermittent porphyria. Clin. Sci. (Lond. Engl. 1979) 1980, 58, 469–476
- Mikirova, N. Clinical Test of Pyrroles: Usefulness and Association with Other Biochemical Markers. Clin. Med. Rev. Case Rep. 2015, 2, 27
- Fryar-Williams S, Strobel JE. Biomarker Case-Detection and Prediction with Potential for Functional Psychosis Screening: Development and Validation of a Model Related to Biochemistry, Sensory Neural Timing and End Organ Performance. Front. Psychiatry 2016, 7, 48
- Jackson JA, Braud M, Neathery S. Urine pyrroles and other orthomolecular tests in patients with ADD/ADHD. J Orthomol Med 2010;25:39
- Die (Krypto-)Pyrrolurie in der Umweltmedizin: eine valide Diagnose? Bundesgesundheitsbl – Gesundheitsforsch – Gesundheitsschutz 2007; 50: 1324-1330
- Voisey J, Swagell CD, Hughes IP, Connor JP, Lawford BR, Young RM, Morris CP. A polymorphism in the dysbindin gene (DTNBP1) associated with multiple psychiatric disorders including schizophrenia. Behav. Brain Funct. 2010, 6, 41
- Voisey J, Swagell CD, Hughes IP, Lawford BR, Young RM, Morris CP. Analysis of HapMap tag-SNPs in dysbindin (DTNBP1) reveals evidence of consistent association with schizophrenia. Eur. Psychiatry 2010, 25, 314–319
- Wockner LF, Noble EP, Lawford BR, Young RM, Morris CP, Whitehall VLJ, Voisey J. Genome-wide DNA methylation analysis of human brain tissue from schizophrenia patients. Transl. Psychiatry 2014, 4, e339
- Wockner LF, Morris CP, Noble EP, Lawford BR, Whitehall VLJ, Young R, Voisey J. Brain-specific epigenetic markers of schizophrenia. Transl. Psychiatry 2015, 5, e680
- Rambaud V, Marzo A, Chaumette B. Oxidative Stress and Emergence of Psychosis. Antioxidants 2022, 11, 1870
- Rothe C, Koszycki D, Bradwejn J, King N, Deluca V, Tharmalingam S, Macciardi F, Deckert J, Kennedy JL. Association of the Val158Met Catechol O-Methyltransferase Genetic Polymorphism with Panic Disorder. Neuropsychopharmacology 2006; 31, 2237–2242
- Zhang X, Gainetdinov RR, Beaulieu JM, Sotnikova TD, Burch LH, Williams RB, Schwartz DA, Krishnan KRR, Caron MC. Loss-of-Function Mutation in Tryptophan Hydroxylase-2 Identified in Unipolar Major Depression Neuron 2005; 45, 11–16
- McDermotta R, Tingleyb D, Cowdenc J, Frazzettod G, Johnson DDP. Monoamine oxidase A gene (MAOA) predicts behavioral aggression following provocation. PNAS 2009; 106 (7): 2118 –2123