Publication Information
ISSN 2691-8803
Frequency: Continuous
Format: PDF and HTML
Versions: Online (Open Access)
Year first Published: 2019
Language: English
| Journal Menu |
| Editorial Board |
| Reviewer Board |
| Articles |
| Open Access |
| Special Issue Proposals |
| Guidelines for Authors |
| Guidelines for Editors |
| Guidelines for Reviewers |
| Membership |
| Fee and Guidelines |
 |
Bioefficacy of the predatory reduviids Rhynocoris kumarii on the hemipteran pests of ––cotton, Dysdercus cingulatus, and Phenacoccus solenopis
Muthupandi M1* Kabilan M1 & Manimaran A2
1Entomology Research Institute, Loyola College (Autonomous), Chennai – 600 034, Tamil Nadu, India
2Department of Advanced Zoology and Biotechnology, Government Arts College for Men, Nanthanam
Received Date: January 06, 2025; Accepted Date: February 10, 2025; Published Date: February 18 2025;
*Corresponding author: Muthupandi M, Entomology Research Institute, Loyola College (Autonomous), Chennai – 600 034, Tamil Nadu, India. Email: muthupandi@loyolacollege.edu
Citation: Muthupandi M, Kapilan M, Manimaran A (2025); Bioefficacy of the predatory reduviids Rhynocoris kumarii on the hemipteran pests of ––cotton, Dysdercus cingulatus, and Phenacoccus solenopis, Enviro Sci Poll Res and Mang: ESPRM-161
DOI: 10.37722/ESPRAM.2025101
Abstract
Mealy bugs and Red cotton bugs cause severe damage to cotton. Biological control may provide an affordable and sustainable option for the reduction of these losses. Functional response and stage preference are the tools employed to evaluate the efficiency of a predator. We studied the bioefficacy of 48 hours starved Rhynocoris kumarii third, fourth and fifth stadium and adult in petri dishes (9.5 cm X 1.3 cm) with cotton leaves. The stage preference was evaluated from the third instar to adult of R. kumarii on all the nymphal stages of Dysdercus cingulatus and Phenacoccus solenopis. Invariably, the third instar of D. cingulatus and P. solenopis were preferred by all the tested life stages of R. kumarii. The functional response of R. kumarii exhibited Holling’s type II curvilinear decelerating response where a positive correlation was obtained between the prey density and the number of prey consumed by the predator. The number of D. cingulatus killed by R. kumarii were 37.4, 40.6, 39, 31.4 preys / predator / day, attack rate wasn0.63, 0.68, 0.65, 0.52 preys / predator, searching time was -2.675, -4.196, -1.535, -1.535 for the third, fourth, fifth and adult predators respectively. With reference to the consumption of P. solenopis, the third instar of R. kumarii consumed the maximum number of prey (5.0 prey / predator / day) than fourth instar (3.55 preys / predator / day) and fifth instar (3.50 preys / predator / day). The rate of attack of adult female predator was quite low (1.50 preys / predator / day) but fairly consistent. From laboratory observation, it can be concluded that the third to fifth nymphal instars of R. kumarii can be used for the management of both P. solenopis and D. cingulatus in cotton fields.
Keywords: Bioefficacy; Dysdercus cingulatus; Functional response; Phenacoccus solenopis; Rhynocoris kumarii; Stage preference
Introduction
Reduviidae constitute an important group of predatory insects in South India. It is the largest family of predatory land heteropterans and many of its members are found to be potential predators of a number of insect pests (Ambrose 1999; 2000; 2003). Since they are polyphagous, they are not effective on specific pests, but they are valuable predators in situations where a variety of insect pests occur (Ambrose et al. 2009). Predation upon a wide array of insects suppresses the efficacy of any organism which is a prerequisite for its utilization as a biological control agent. The description of a predator’s instantaneous feeding rate or predatory impact, as a function of prey density is its functional response (Ghosh and Chandra 2011). The functional response of a predator is a key factor in regulating the population dynamics of predator prey systems in any ecosystem. It describes the rate at which a predator kills its prey at different prey densities and can thus determine the efficiency of a predator in controlling prey populations in biological control programmes in laboratory conditions or in field survey (Oaten and Murdoch 1975). The functional response of a natural enemy offers a good conceptual framework for understanding the action of agents in inundative releases in the field also (Waage and Greathead 1988). The functional response curves can be differentiated by evaluating the parameters such as attack rate and handling time. The attack rate estimates the steepness of the increase in the rate of predation with increasing prey density and handling time is very useful in estimating the satiation threshold. Arthropod predators generally display one of the three types of functional responses. The functional response curves may represent an increasing linear relationship (Type I), a decelerating curve (Type II) or a sigmoidal relationship (Type III) (Pervez and Omkar 2005).
Rhynocoris kumarii (Ambrose and Livingstone) is a harpactorine reduviid, whereas Dysdercus cingulatus and Phenacoccus solenopis are hemipteran pests. Both the predator and pest are widely distributed in tropical rain forest, scrub jungle and semi arid zone.
The present study was designed to evaluate the predatory efficiency through stage preference and functional response of R. kumarii on D. cingulatus and P. solenopis life stages with body weight of the prey and predator, because of the lack of literature and report available with respect to the specific distribution of D. cingulatus and P. solenopis in forest ecosystem and their control management.
Materials and method
The pests D. cingulatus and P. solenopis were collected from the forest ecosystem in Tirunelveli (8.7300° N, 77.7000° E) and Kanyakumari (8.0780° N, 77.5410° E) districts of Tamilnadu and were maintained under laboratory conditions (Temperature 31-32ºC; RH 75-80%; Photoperiod 11-13 hours) in a plastic trough (22x10x14 cm) on soaked cotton seed. The nymphs and adults of R. kumarii were also collected from forest ecosystems in Tirunelveli and Kanyakumarii districts of Tamil Nadu and were reared in the laboratory (Temperature 31-32ºC; RH 75-80%; photoperiod 11-13 hours) in separate plastic containers (8x6x4 cm) on Corcyra cephalonica (stainton) larvae (Sahayaraj 2007; Sahayaraj and Asha 2010) and Spodoptera litura.
The Stage preference of both the pests was studied by exposing all the stages (1st to adult) of prey separately in the petri dish to the predator. Observations were made for 24 hours on the successful predation and the stages of prey preferred. This experiment was repeated five times.
The functional response of predator was assessed separately at six densities viz, 1, 5, 10, 20, 40 and 60 prey / predator. The prey was introduced into the petri dish (9.5 cm dia x 1.3 cm height) with cotton leaf. After selection of the prey the predators were released. Approaching time and handling time were observed. After 24 hours, the numbers of prey consumed or killed were counted. In the present study the disk equation of Holling (1959) was used to describe the functional response of R. kumarii. Prey density (x), total number of prey killed in the given period of time (y), and the maximum prey consumed (k) were recorded. Prey attack ratio (y/x), rate of discovery per unit of searching time (a=y/x/Ts), time spent by the predator in searching prey (Ts), total time when prey was exposed to the predator (Tt), the maximum number of prey consumption (k), and the time spent for handling each prey by the predator (b=Tt/k) were calculated using disc equation (Sahayaraj and Asha 2010). Mean number of prey killed by a predator as a function of prey density was subjected to correlation in SPSS software and significance was expressed at 0.05% level.
Result
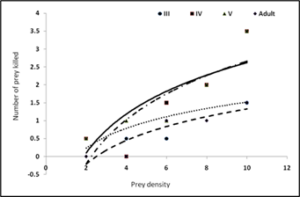
Third, fourth, fifth and adults of R. kumarii responded to the increasing prey density by killing higher number of prey (R) than it killed at lower prey density. This was confirmed by positive correlation recorded between the prey density and the number of prey killed during 24 hours (r=0.09, 0.99, 0.97, 0.75 for third, fourth, fifth and adult of D. cingulatus and r=0.97, 0.98, 0.94, 0.98 for P. solenopis respectively as seen in table 1 and 2). Functional response revealed that prey consumption by R. kumarii increased with increase in prey density, which exemplifies Hollings type II functional response. This was further confirmed by the positive correlation, obtained between the prey density and prey killed irrespective of the predator life stages (third, fourth, fifth instars and adults) tested. The maximum predation expressed by ‘K’ value was always found restricted to high prey density. The weight of the predator and the prey weight were considered for functional response and stage preference. The weight of the third, fourth and fifth instar and adult of R. kumarii were 6.63 mg, 14.18 mg, 32.71 mg and 21 mg respectively. All these predator stages preferred the third instar of D. cingulatus (11.32 mg) and P. solenopis (000). This is due to the high amount of haemolymph and easy handling of the prey helped in killing and feeding. Time spent by the predator for searching (ys) and handling (b) the prey was found to increase gradually as the predator grew older. The same trend was observed for rate of discovery (a). From the life stages observed, maximum and minimum attack ratios were observed in fourth nymphal instar and adult of R. kumarii respectively. It can be seen from Table 1 that the predator spent less time for searching its prey at maximum density. The fifth instar of R. kumarii shows highest rate of discovery (9.067) at 40 prey density.
| Life Stages | Prey Density | No of prey consumed | Consumed ratio (y/x) | Handling (by) | Days searching (Ts=1-by) | Prey Y’=a(1-by) | Predicted attack Y’/x | Max (Y) | B=by/y | Rate of discovery (a=y/x/Ts) |
| Third instar | 1 | 01 | 1.0 | 0.09 | 0.37 | 0.99 | 0.99 | 0.09 | 1.098 | |
| 5 | 3.8 | 0.76 | 0.281 | 0.719 | 0.75 | 0.15 | 0.0739 | 1.057 | ||
| 10 | 5.6 | 0.56 | 0.347 | 0.653 | 0.56 | 0.056 | 0.0619 | 0.857 | ||
| 20 | 14.2 | 0.71 | 1.448 | -0.448 | 0.71 | 0.035 | 0.1019 | -1.584 | ||
| 40 | 24 | 0.6 | 3.024 | -2.024 | 0.59 | 0.0147 | 0.126 | -0.296 | ||
| 60 | 37.5 | 0.625 | 3.675 | -2.675 | 0.623 | 0.0104 | 37.5 | 0.098 | -0.233 | |
| Fourth instar | 1 | 1 | 1 | 0.048 | 0.952 | 0.99 | 0.99 | 0.048 | 1.0504 | |
| 5 | 3 | 0.6 | 0.342 | 0.658 | 0.59 | 0.12 | 0.114 | 0.9118 | ||
| 10 | 6 | 0.6 | 0.516 | 0.484 | 0.59 | 0.06 | 0.086 | 1.2396 | ||
| 20 | 14.4 | 0.72 | 1.238 | -0.238 | 0.12 | 0.0006 | 0.085 | 0.4911 | ||
| 40 | 27.4 | 0.685 | 2.466 | -1.466 | 0.68 | 0.017 | 0.09 | -0.4672 | ||
| 60 | 40.6 | 0.677 | 5.196 | -4.196 | 0.676 | 0.112 | 40.6 | 0.13 | -0.1613 | |
| Fifth instar | 1 | 1 | 1 | 0.07 | 0.93 | 0.99 | 0.99 | 0.07 | 1.0752 | |
| 5 | 3.8 | 0.76 | 0.220 | 0.78 | 0.759 | 0.151 | 0.0578 | 0.9743 | ||
| 10 | 6.8 | 0.68 | 0.462 | 0.538 | 0.679 | 0.067 | 0.0679 | 1.2639 | ||
| 20 | 13.6 | 0.68 | 0.925 | 0.075 | 0.679 | 0.034 | 0.680 | 9.0666 | ||
| 40 | 30.4 | 0.76 | 2.188 | -1.188 | 0.759 | 0.018 | 0.0719 | -0.6397 | ||
| 60 | 39 | 0.65 | 2.535 | -1.535 | 0.649 | 0.010 | 39 | 0.065 | -0.4234 | |
| Adult | 1 | 1 | 1 | 0.05 | 0.95 | 1.000 | 1 | 0.05 | 1.053 | |
| 5 | 3.6 | 0.72 | 0.55 | 0.45 | 0.72 | 0.144 | 0.153 | 1.6 | ||
| 10 | 7.8 | 0.78 | 0.067 | 0.933 | 0.056 | 0.0005 | 0.0008 | 0.836 | ||
| 20 | 14 | 0.7 | 0.091 | 0.91 | 0.699 | 0.0349 | 0.0006 | 0.769 | ||
| 40 | 25.8 | 0.645 | 0.0542 | 0.95 | 0.644 | 0.0161 | 0.0210 | 0.678 | ||
| 60 | 31.4 | 0.523 | 0.039 | 0.961 | 0.522 | 0.0008 | 31.4 | 0.0001 | 0.544 |
Table 1: Functional response of Rhynocoris kumarii on Dysdercus cingulatus.
| Life Stages | Prey Density | No of prey consumed | Consumed ratio (y/x) | Handling (by) | Days searching (Ts=1-by) | Prey Y’=a(1-by) | Predicted attack Y’/x | Max (Y) | B=by/y | Rate of discovery (a=y/x/Ts) |
| Third instar | 1 | 0.50 | 0.50 | 0.72 | 0.28 | 0.49 | 0.49 | 1.78 | ||
| 2 | 1.50 | 0.75 | 3.56 | -2.56 | 1.48 | 0.74 | 0.29 | |||
| 4 | 2.30 | 0.57 | 1.76 | -0.76 | 2.28 | 0.57 | 0.75 | |||
| 6 | 2.70 | 0.45 | 2.77 | -1.77 | 2.65 | 0.44 | 0.25 | |||
| 8 | 4.90 | 0.61 | 3.35 | -2.35 | 4.70 | 0.58 | 0.25 | |||
| 10 | 5.00 | 0.50 | 2.91 | -1.91 | 4.90 | 0.49 | 5.00 | 0.58 | 0.26 | |
| Fourth instar | 1 | 0.75 | 0.75 | 1.94 | -0.94 | 0.75 | 0.75 | 1.08 | ||
| 2 | 1.60 | 0.80 | 2.57 | -1.57 | 1.59 | 0.79 | 0.52 | |||
| 4 | 2.25 | 0.56 | 2.42 | -2.84 | 2.20 | 0.55 | 0.47 | |||
| 6 | 2.60 | 0.43 | 1.27 | 0.51 | 2.57 | 0.42 | 1.10 | |||
| 8 | 3.45 | 0.35 | 2.27 | -1.27 | 3.34 | 0.33 | 4.07 | |||
| 10 | 3.55 | 0.44 | 2.01 | -1.12 | 3.39 | 0.43 | 3.55 | 0.57 | 2.35 | |
| Fifth instar | 1 | 0.60 | 0.60 | 0.18 | 0.82 | 0.59 | 0.59 | 0.73 | ||
| 2 | 1.80 | 0.90 | 2.59 | -1.59 | 1.78 | 0.89 | 0.56 | |||
| 4 | 2.20 | 0.55 | 0.32 | 0.68 | 2.17 | 0.54 | 0.80 | |||
| 6 | 2.50 | 0.41 | 1.21 | -0.21 | 2.45 | 0.40 | 1.95 | |||
| 8 | 3.30 | 0.41 | 1.45 | -0.45 | 1.52 | 0.19 | 0.91 | |||
| 10 | 5.50 | 0.55 | 2.82 | -1.82 | 5.46 | 0.54 | 5.50 | 0.51 | 0.30 | |
| Adult | 1 | 0.40 | 0.40 | 0.26 | 0.74 | 0.39 | 0.39 | 0.58 | ||
| 2 | 0.95 | 0.47 | 0.51 | 0.48 | 0.94 | 0.46 | 1.07 | |||
| 4 | 1.15 | 0.28 | 0.56 | 0.43 | 1.13 | 0.27 | 0.78 | |||
| 6 | 1.60 | 0.26 | 0.67 | 0.33 | 1.57 | 0.26 | 0.86 | |||
| 8 | 1.35 | 0.16 | 0.58 | 0.41 | 1.30 | 0.16 | 0.78 | |||
| 10 | 1.60 | 0.16 | 0.63 | 0.36 | 1.58 | 0.15 | 1.90 | 0.38 | 0.89 |
Table 2: Functional response of Rhynocoris kumarii on Phenacoccus solenopis.
Discussion
Rhynocoris kumarii (Ambrose and Livingstone) is an important hunter reduviid. Research on reduviids show that they are distributed in Marunthuvalmalai (Vennison and Ambrose 1990) Sungankadai (Kumar 1993), Godayan (Ambrose and Sahayaraj 1991) Courtallam (Edwin and Ambrose 1996), Azhagarmalai (Kumarasami and Ambrose 1991-1992), Kalakad, Petchiparai, Papanasam and Sivagiri. But in Kalakad, Petchiparai, Papanasam, Sivagiri ecosyatems, they haven’t been recorded before.
Rhynocoris kumarii is found to predate on C. compressus, H. armigera, (Ambrose 1980, 1987), P. succinta (Ambrose and Livingstone, 1987), M. pustulata (Kumaraswami 1991), D. cingulatus (Ambrose 1995), M. indica, P. gossypiella, S. lilana, E. scintillians (Ambrose and Claver 1995a, 1995b), P. solenopis, E. merinone, C. cephalonica, E. insulana, E. vitella, O. hyalinipennis, E. mollifera, Tribolium confusum Duv, Papillo demoleus Linn., R. clavatus, C. gibbosa, D. indicus, E. atomosa (Ambrose 1999). The above mentioned reports have mentioned only the preference of the pest. In this present study, the density of the prey was high when compared with the previous studies. The weight of the prey and predator was considered.
Dysdercus cingulatus and Phenococcus solenopis are hemipteran pests. They have the following natural enemies namely, R. marginatus (Ambrose 1999), Corianus nodulosus (Sahayaraj 1997), Endochus umbrinus (Sahyaraj 1991), R. kumarii (Ambrose 1995), Ectomocoris tibialis (Ambrose 1999), Neara vividula parasitoid, Antilochus coquebertii (Yunus and Ho 1980; Zaidi and Zaheer 1985), Mite Hemipterotarseius sp. and the nematode Stenesnema carcocapsae.
D. cingulatus and P. solenopis infest many of the forest plants. So, we investigated the control strategies of these pests in R. kumarii, because R. kumarii was found to be present in all the plants where D. cingulatus and P. solenopis were present. These predators are polyphagus bugs that feed mainly on leaf feeding larvae of Lepidoptera, Isoptera, Coleoptera and Hemiptera. Numerous studies have been reported on the predatory potential of reduviids (Sahayaraj 1994a; 1994b; 1995; 2000; 2006; Sahayaraj and Sivakumar 1995; Sahayaraj and Ambrose 1994), but information regarding the predatory potential against D. cingulatus and P. solenopis has been minimum. Predatory efficiency of these reduviids exhibited type II functional response in A. cranivora and also other reduviids (Sahayaraj 1991; Sahayaraj and Raju 2006). Here, the results show the inversely proportional relationship between attack and prey level. It is presumed that the predator required less time to search the prey and more time on non-searching activities at higher prey densities, which in turn might have caused perceptive decline in the attack rate until hunger was established. Moreover, higher prey density also result in reduction in unsuccessful attacks of a predator on a prey, as there are less chances of escape when compared to those in scarce prey density, where there are more chances for the prey to escape from the predator (Sahayaraj 2006). Hunger and time spent by the predator in searching and handling the prey can affect the prey consumption and eventual preparation of the predator for further search.
In this study, third, fourth, fifth instars and adult prefer the third instar of D. cingulatus and P. solenopis. It may be due to the haemolymph content of the pest and its mobility. From this study, the predator and prey ratio of third, fourth, fifth instar and adult of R. kumarii was 1:38, 1:41, 1:39, and 1:31 for D. cingulatus and 1:5, 1:3.55, 1:3.5 and 1:2.3 for P. solenopis respectively. According to our study, the fourth instar and the third instar of R. kumarii have the potential to suppress the population of D. cingulatus and P. solenopis respectively. The correlations between third, fourth, fifth and adult of R. kumarii with four instars of D. cingulatus are 0.999, 1, 0.995, and 0.989 and with P. solenopis are 0.97, 0.98, 0.94, and 0.98. This is significant at 0.01 levels. Multiple comparisons show that the lowest predensity having significant values at 0.05 levels, density 1 has 0.504. The pests D. cingulatus and P. solenopis are distributed worldwide and they damage cotton crops. In our study we collected these pests in Cordispermum helicabum (Scorbularaceae), Pavonia odarata (Malvaceae), Leucus aspera (Lamiaceae). These plants have considerable medicinal value. D. cingulatus and P. solenopis are polyphagous. Thus, they are found to shift to other plants when one host plant gets washed out. Hence the control of these pests becomes indispensable in order to protect the medicinal plants.
Acknowledgement: The authors wish to express their heartfelt gratitude to Loyola College and its authorities for Institutional facilities.
References
- Ambrose DP (1980) Bioecology, Ecophysiology and Ethology of Reduviids (Heteroptera) of the Scrub Jungles of Tamil Nadu, India. Ph.D. Thesis, University of Madras, Madras, India.
- Ambrose DP (1987) Assassin bugs of Tamil Nadu and their role in biological control (Insecta: Heteroptera: Reduviidae), 16-28. In: Joseph KJ and Abdurahiman UC (Eds.). Advances in Biological Control Research in India. Printex Ltd., Calicut.
- Ambrose DP (1995) Reduviids as predators: Their role in biological control. In:Biological control of social forests and plantation crops insects, (T.N. Ananthakrishnan, New Delhi: Oxford & IBH Publishing Co. Ltd.,) (Ed.) 153-170.
- Ambrose DP (1999) Assasin Bugs. Enfield New Hampshire, USA.
- Ambrose DP (2000) Substrata impact on mass rearing of the reduviid predator Rhynocoris kumarii, Ambrose and Livingstone (Heteroptera:Reduviidae). J. Znt. Res. 24:337-342.
- Ambrose DP (2003) Biocontrol potential of assassin bugs (Hemiptera:Reduviidae) J. Exp. Zool. India. 6:1-44.
- Ambrose DP, Claver MA (1995a) Food requirement of Rhynocoris kumarii Ambrose and Livingstone (Heteroptera: Reduviidae). Journal of Biological Control 9: 47 -50.
- Ambrose DP, Claver MA (1995b) Functional response of Rhynocoris fuscipes (Fabricius) (Heteroptera:Reduviidae) to Riptortus clavatus (Thin bag) (Hemiptera: coreidae). J. Biological control. 9:74-77.
- Ambrose DP, Livingstone D (1987) Biology of a new harpactorine assassin bug Rhynocoris kumarii (Hemiptera: Reduviidae) in South India. Journal Soil biology and Ecology 7: 48-58.
- Ambrose DP, Rajan JS, Nagarajan K, Singh VJ Krishnan SS (2009) Biology, Behaviour and Functional Response of Sphedanolestes Variabilis Distant (Insecta : Hemiptera : Reduviidae : Harpactorinae ), a potential predator of Lepidopteran pests. Croat. 13:33-44.
- Ambrose DP, Sahayaraj K (1991) Camouflaging behaviour and the effect of decamouflaging on behaviour and development of Acanthaspis pedestris Stal in solitary and crowded conditions (Heteroptera: Reduviidae). Zoo. Muslim, Berlin 67: 325-337.
- Edwin J, Ambrose DP (1996) Population dynamics of Reduviid fauna (Insecta: Heteroptera: Reduviidae) in Courtallam tropical rain forest, Western Ghats. Biological and cultural control of Insect pests, an Indian scenario (ed.) Tirunelveli India, Adeline Publishers. 94-102.
- Ghosh A, Chandra G (2011) Functional responses of Laccotrephes griseus (Hemiptera: Nepidae) against Culex quinquefasciatus (Diptera: Culicidae) in laboratory bioassay. J Vector Borne Dis. 48:72-7.
- Holling CS (1959) The functional response of invertebrate predators of prey density Mem. Ent. Soc. Canada. 48.
- Kumaraswami NS (1991) Bioecology and ethology of chosen predatory assassin bugs and their potential in biological control (Insecta: Heteroptera: Reduviidae). PhD Thesis, Madurai Kamaraj University, Madurai.
- Kumaraswami NS, Ambrose DP (1991) Bioecology and ethology of Edocla annulata Distant (Hemiptera: Reduviidae: Acanthaspidinae) from South India. Journal of Soil Biology (17): 33-42.
- Oaten A, Murdoch W (1975) Functional response and stability in predator prey systems. The American Naturalist 109:289-298.
- Pervez A, Omkar (2005) Functional responses of coccinellid predators: An illustration of a logistic approach. Journal of Insect Science. 5:5.
- Sahayaraj K (1991) Bioecology, ecophysiology and ethology of chosen predatory hemipterans and their potential in biological control. Ph.D. Thesis. Madurai Kamaraj University, Madurai. 162-190.
- Sahayaraj K (1994a) Bio-control potential evaluation of the reduviid predator Rhynocoris marginatus (Fabricius) to the serious groundnut pest Spodoptera litura (Fabricius) by functional response study. Fresenius Environmental Bulletin 3: 546-550.
- Sahayaraj K (1994b) Capturing success by reduviid predators Rhynocoris kumarii and marginatus on different age groups of Spodoptera litura, a polyphagous pest (Heteroptera: Reduviidae). Journal of Ecobiology 6: 221-224.
- Sahayaraj K (1995) Biocontrol potential and development of reduviid predator Acanthaspis pedestris Stal on Odontotermes sp: a serious groundnut pest. International Archive Newsletter 15:59-60.
- Sahayaraj K (2000) Evaluation of biological control potential of Rhynocoris marginatus (Fab) on four groundnut pests under laboratory condition. International Archive Newsletter. 20:72-74.
- Sahayaraj K (2006) Ecological adaptive features of Hunter Reduviids (Insecta: Heteroptera: Reduviidae Latreille 1807) and their biological control potential. In:Prespectives in Animal Ecology and Reproduction (III) Eds. VK Guptha and Anil K Verma, 3:25- 41.
- Sahayaraj K (2007) Ecotypic variation in the biology of Acanthaspis quinquepinosa Fabricius 1781 (Hemiptera: Reduviidae: Reduviinae) from peninsular India. Egyptian journal of biology 9:53-59.
- Sahayaraj K, Ambrose DP (1994) Host preference and functional response of a reduviid predator Acanthaspis pedestris Stal to four cotton pests. Journal of biological control 8:23-26.
- Sahayaraj K, Asha A (2010) Biological control potential evaluation of Rhynocoris kumarii Ambrose and Livingstone on Aphis craccivora (Koch). Indian Journal of Agricultural Sciences, 44: 281 – 287.
- Sahayaraj K, Raju G (2006) Assessing the intraguild predation of Rhynocoris marginatus (Fabricious) (Heteroptera: Reduviidae: Harpactorinae) and Menochilus sexmaculatus (Fab.) of cotton pest Aphis craccivora. Hexapoda 13: 62 – 65.
- Sahayaraj K, Sivakumar K (1995) Groundnut pest and pest stage preference of a reduviid predator Rhynocoris kumarii Ambrose and Livingstone (Heteroptera: Reduviidae). Fresnius Environmental Bulletin 4: 263-269.
- Vennison SJ, Ambrose DP (1990) Biology and behaviour of Sphedanolestes signatus Distant (Insecta: Heteroptera: Reduviidae) a potential predator of Helopeltis antonii Uttar Pradesh Journal of Zoology. 10:30-43.
- Waage JK, Greathead DJ (1988) Biological control: challenges and opportunities. Philos. Trans. R. Soc. Lond. B 318:111-128.
- Yunus A, Ho TH (1980) List of economic pests, host plants, parasites and predators in West Malaysia (1920-1978). Ministry of Agriculture, Malaysia, Bulletin 153: 538.
- Zaidi, Zaheer S (1985) Proteolytic activity in the gut of red cotton bug, Dysdercus cingulatus Fabr. and its predator, Antilochus conquebertii (fabr.) (Heteroptera: Pyrrhocoridae)
Figure 1. Bioefficacy of Rhynocoris kumarii third instar on D. cingulatus third instar.
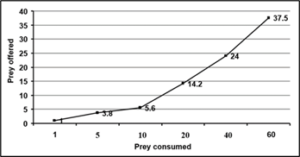
Figure 2: Bioefficacy of Rhynocoris kumarii fourth instar on D. cingulatus third instar.
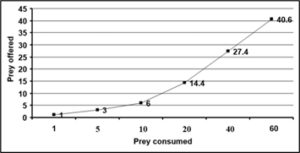
Figure 3: Bioefficacy of Rhynocoris kumarii fifth instar on D. cingulatus third instar.
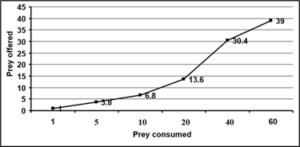
Figure 4: Bioefficacy of Rhynocoris kumarii adult on D. cingulatus third instar.
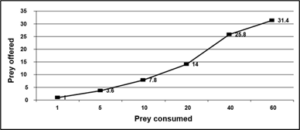
Figure 5: Type II functional response curve of Rhynocoris kumarii third, fourth and fifth nymphal instar and adults preying upon different densities of Phenacoccus solenopsis.